In Jay Stevens’ classic social history of LSD, Storming Heaven: LSD and the American Dream, the author discusses how Ken Kesey — of Merry Pranksters fame and author of One Flew Over the Cuckoo’s Nest — volunteered for a “medical experiment” run by MK-ULTRA-connected physician Leo Hollister in which he was given a variety of “psychotomimetic” drugs:
“The first drug Hollister gave Kesey was psilocybin… In the ensuing weeks, Kesey was dosed with LSD, the super-amphetamine IT-290, and Ditran, which was a synthetic relative of belladonna. Kesey always knew it was Ditran because the fibers on his blanket would turn into a field of thorns and then he'd throw up.”
Psilocybin and LSD are obviously familiar, but IT-290 likely much less so. I’ll discuss Ditran another time — it’s an interesting subject on its own — but this so-called “super-amphetamine” immediately caught my attention when reading this section. It turns out that IT-290 was the moniker given to a stimulant being developed as an antidepressant in the 1960s by Sandoz. The Upjohn company were also developing the same molecule, as U-14, and, in Russia, it actually made it to market as an antidepressant known as Indopan. Nothing like a bit of Cold War intrigue to get the pharmaceutical juices flowing. Unfortunately, IT-290 isn’t as “super” as claimed and isn’t even an amphetamine. But it is, in other ways, quite intriguing.
From its molecular structure alone, IT-290, more commonly known as alpha-methyltryptamine (AMT) appears fairly unremarkable: it’s simply tryptamine with a methyl group attached adjacent to the amine group (in the alpha position, hence the name). However, what makes AMT interesting is precisely this simplicity and how it relates to tryptamine which is, of course, the basic molecule upon which all psychedelic tryptamines are elaborated.
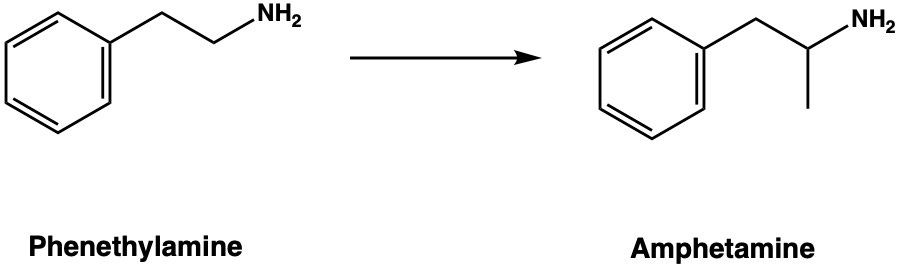
Going from tryptamine to AMT is equivalent to going from phenethylamine to amphetamine, which is alpha-methyl-phenethylamine (otherwise known as phenylisopropylamine). In fact, this is precisely how one defines an amphetamine, as a phenethylamine with a methyl group in the alpha position, and is probably why AMT was referred to as a “super-amphetamine”. But it’s not an amphetamine, by definition. So, what’s the significance of this alpha-methyl group?
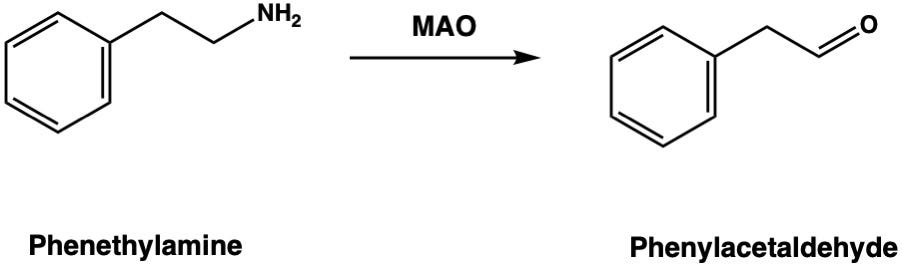
Phenethylamine itself is usually reported as being completely inactive orally, since it’s rapidly degraded by monoamine oxidase (MAO) as soon as it enters the body. MAO cleaves the crucial amine group from the side chain and converts it to a carboxyl group, rendering the molecule inactive. If it survived, it would probably be a stimulant and, according to Andrés Gómez Emilsson of the Qualia Research Institute, it does indeed have such effects if you swallow enough to overwhelm MAO:
“You can get really high with it if you take 500mg+. I did that a few times in grad school… It's very stimulating, weird, a bit MDMA like in some ways, but also full of anxiety… It's a *rush* that lasts about 20 minutes…”
Adding the methyl group makes it a poor substrate for MAO, which is why amphetamine is much more active orally at much lower doses and lasts several hours (rather than 20 minutes or so).
In fact, adding an alpha-methyl group to a psychoactive phenethylamine to form the amphetamine analogue is a fairly reliable way of making a more potent and longer-lasting drug. Take 2C-B (4-bromo-2,5-dimethoxyphenethylamine), for example, which is active 10-25 mg range and lasts for 4-8 hours. Adding the alpha-methyl generates DOB (2,5-dimethoxy-4-bromoamphetamine), which is fully active between 1 and 3 mg and lasts for up to 30 hours! Or, we can also take the relatively unknown MDPEA (3,4-methylenedioxyphenethylamine), which is completely inactive and, yet, add that alpha-methyl and we get MDA (3,4-methylenedioxyamphetamine), a popular psychedelic amphetamine active in the 80-160 mg range and lasting 4-6 hours. That alpha-methyl makes all the difference!
In my previous post on the mechanism of amphetamines, I discussed how their euphoric, stimulating, and empathogenic effects primarily results from them interfering with protein pumps controlling the re-uptake of noradrenaline, dopamine, and serotonin from the synaptic cleft back into the neuron, and even causing these pumps to shift into reverse and actively pump these neuromodulators out of the neuron. The net result is an increase in the concentration of these neuromodulators within the cortex (and thus an increase in their effect). If you haven’t read it, I suggest you do so:
Psychedelic Amphetamines, Speedy Amphetamines, Entactogenic Amphetamines...
One of the most fascinating aspects of neuropharmacology, for me at least, is how closely related molecules, perhaps differing only by the presence or absence of a methyl group (think DMT vs 5-MeO-DMT), can have dramatically different effects on the brain and consciousness. Whilst we understand much about the way molecules interact with receptors, how t…
The psychedelic effects of certain substituted amphetamines likely results from additional activity at the serotonin 5HT2A receptor (the site of action of the classic psychedelics). The balance of stimulating, euphoric, empathogenic, and psychedelic activity of a particular amphetamine depends upon the relative activity at these transporters and the 5HT2A receptor.
Tryptamine itself suffers the same metabolic issue as phenethylamine: as soon as it enters the body, it’s rapidly degraded. However, since it has a high affinity for the serotonin 5HT2A receptor, it’s possible that it might be psychedelic if it escaped MAO degradation and made it safely into the brain. Whilst oral and bolus injection of tryptamine are wholly (and unsurprisingly) inactive, a study way back in 1970 found that if up to 277mg of tryptamine was infused intravenously over several minutes — any longer and the subjects began vomiting — minor perceptual changes occurred, although becoming “more aware of the experimental setting” and complaining of a “heaviness” is a far cry from any half-decent LSD experience (LINK). Most of the effects the authors describe were unpleasant bodily effects, such as dizziness, sweating, and “numbness of the limbs”. This is clearly not a molecule of any particular psychoactive interest, but perhaps shows that, if you manage to get enough tryptamine into the bloodstream to overwhelm the MAO system such that a small proportion makes it into the brain, there might be some psychoactivity there.
So, if we want to convert tryptamine to a psychoactive molecule, adding an alpha-methyl group to form AMT might just do the trick…
AMT was first synthesised in 1947, but its psychoactive properties weren’t reported until more than a decade later. One of the first papers describing its effects was published in 1960 by the same Leo Hollister mentioned by Jay Stevens, which describes administering AMT, as well as psilocybin and Ditran, to 16 subjects, presumably including Ken Kesey (LINK).
Depending on the dosage, the effects of AMT were quite variable: Low (5-10 mg) doses elicited a relatively subtle mood-lifting effect, shifting up a gear to outright euphoria at higher (20 mg) doses and, at very high (30+ mg) doses, a pronounced psychedelic effect emerged, although it’s generally described as being much less deep and profound than LSD or psilocybin. This is not entirely dissimilar to MDMA, which also has increasing psychedelicity as the dosage increases, likely owing to activity at 5HT2A receptors, a property shared with AMT. Also similar to MDMA, AMT is a potent inhibitor of both noradrenaline and serotonin re-uptake, explaining its neurostimulatory and empathogenic effects. However, it’s considerably more potent than MDMA at inhibiting dopamine re-uptake, with about the same potency as cocaine and approaching that of methamphetamine, which explains its particularly euphoric effects (LINK). However, unlike MDMA, AMT users are more likely to experience negative psychological and physical effects, particularly at higher doses, including nervousness, anxiety, and irritability, as well as muscle tightness and vomiting. Owing to this body-load, many users find the overall experience to be rather unpleasant, whereas others find AMT to be an enjoyable party drug — it definitely seems to be less reliable than other psychostimulants if you’re looking for a good time (LINK). This unreliability is compounded by a delay of 2-3 hours before the effects kick in, suggesting that an active metabolite might be responsible for its effects, rather than AMT itself. Simple molecule, messy pharmacology. Super-amphetamine? Not so much.
So, how might we improve upon AMT to produce something more reliable, either as a stimulant or psychedelic? Extending the alpha-methyl to an ethyl group produces alpha-ethyltryptamine (AET), which is much less potent than AMT, with active doses in the 100+mg range, but also seems to lack many of the negative side-effects, with a much cleaner feeling of well-being and euphoria without the jitteriness of AMT, as well as being less psychedelic and with a much faster onset (LINK). As such, it was briefly marketed as the anti-depressant “Monase” by Upjohn in the early 1960s, until potentially severe side-effects on white blood cell counts began to appear in some patients and, in 1962, it was promptly discontinued. Recreational use of AET peaked in the 1980s, when it was still unscheduled — the DEA put an end to the fun in 1993 by dropping it into Schedule I.
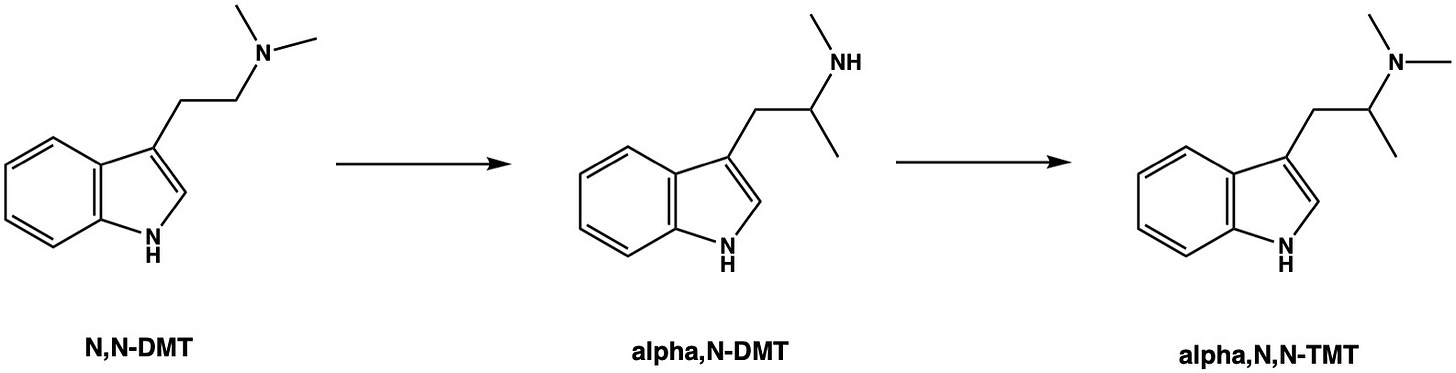
An obvious alternative to playing with the alpha-methyl group is to add a methyl group to the amine nitrogen — this is the equivalent of converting amphetamine to its more potent close cousin, methamphetamine. This yields alpha,N-DMT, which can also be seen as N,N-DMT (i.e. good old Spirit Molecule, DMT) with one of the methyl groups shifted from the amine to the alpha carbon. Unfortunately, rather than potentiating the simulating properties of AMT, adding this amine methyl makes for a rather uninteresting molecule with some mild, generally uncomfortable, stimulation in the 50-100 mg range. We can push this further by adding a second methyl group to the amine, generating the alpha-methylated version of DMT — alpha,N,N-trimethyltryptamine — but this is even less active as a stimulant and certainly isn’t a psychedelic.
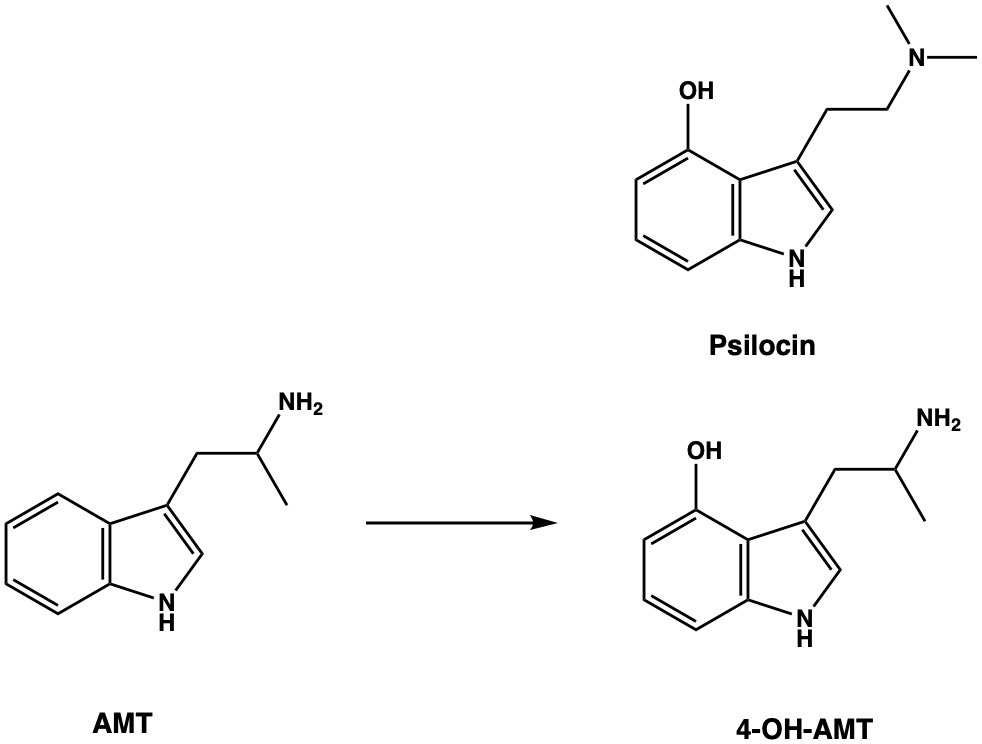
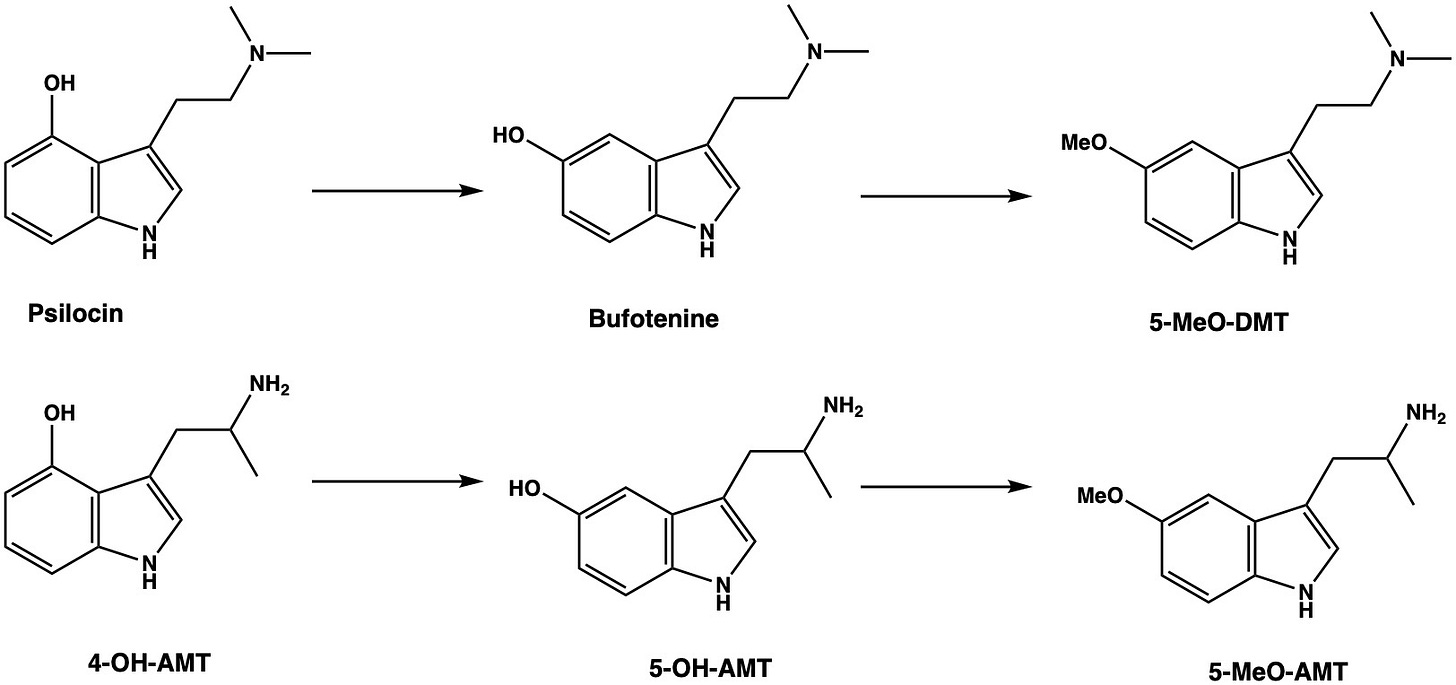
It turns out that we’ll have more luck by shifting the focus from the side chain to the indole ring, where we have numerous options for structural embellishment. An obvious choice is to add a hydroxyl group to the indole 4-position — the same as psilocin — which generates the alpha-methyl analogue of norbaeocystin, 4-OH-AMT.
According to Shulgin:
“It is reported to be markedly visual in its effects, with some subjects reporting dizziness and a depressed feeling. There were, however, several toxic signs at doses of 15 to 20 milligrams orally, including abdominal pain, tachycardia, increased blood pressure and, with several people, headache and diarrhea.”
Again, not particularly interesting or desirable. So, let’s shift the hydroxyl to the 5-position on the indole ring, generating the 5-OH-AMT, which could also be called alpha-methyl-serotonin (alpha-methyl-5HT). This turns out to be a useful analogue of serotonin in scientific studies, since it has similar 5HT receptor binding properties to serotonin (technically, it’s a non-selective serotonin receptor agonist, just like serotonin) but, since it’s a poor substrate for monoamine oxidase, sticks around for much longer. However, unfortunately, it’s not particularly psychoactive and, just like serotonin, is unlikely to readily cross the blood brain barrier. So, again, not a particularly interesting molecule. But we shan’t give up just yet.
We can mask the polarity of the 5-hydroxy group, which limits absorption in the brain, by adding a methyl group, converting it to the methoxy group and generating 5-MeO-AMT — this should cross the blood brain barrier much more easily. This is equivalent to converting bufotenine (5-OH-DMT), which is of disputable psychoactivity and struggles to cross the blood brain barrier, to 5-MeO-DMT, which is a highly potent psychedelic.
Here things become more interesting: this 5-methoxy group is significant since, in 5-MeO-DMT, it forms a hydrogen bond with a serine in the 5HT2A receptor ligand binding site. So, we might expect 5-MeO-AMT to bind similarly and perhaps shift the molecule away from neurostimulation and more towards the psychedelic end of subjective effects. And, indeed, this is exactly what we see: 5-MeO-AMT (also known as Alpha-O), whilst quite stimulating, is also a highly potent psychedelic, fully active in the 2-5 mg range and lasting upwards of 12 hours (dependent on 5HT2A receptor activation, as with other classic psychedelics — LINK). Effects are quite varied, however, with as many users describing an extremely positive experience as those finding the trip more difficult, often owing to a rather heavy body load (including headaches, nausea, and tachycardia) and a distinctly dissociative aspect to the state. It’s also quite easy to overdose, with a number of hospitalisations and possible deaths being reported (LINK). So, overall, it’s hard to make a solid case for this one.
We needn’t stop here, and there are plenty of other alpha-methyltryptamines that we could try. Unfortunately, there aren’t really any particularly interesting or desirable — let alone “super” — ones that have so far been discovered. Even Shulgin seemed to give up on this class after a while and you’ll only find a handful in TIHKAL, none especially remarkable. Making new, interesting, and safe psychedelic drugs without unpleasant or dangerous side-effects is by no means an easy business — although Shulgin created hundreds of new psychoactive molecules, all assayed by his “make ’em and taste ’em” procedure, only a handful really shine. How many great ones can you name?












Hey buddy, I hope all is well. I was wondering if you may be able to do a piece on Nitrous Oxide. There seems to be loads of confusion about the harmfulness of it and even how it interacts with our brain. It also does the most wild thing with psychedelics (4-ACO-DMT and Nitrous anyone?). I would love to know how it interacts with them and any possible harm it can cause. Also love to know more about the right balance because there seems to be knack to mixing them with oxygen to get the proper effects. :) Thanks n peace.
Ive been researching Ken Kesey’s private collection letters from his participation at the Stanford Va Hospital drug experience in Menlo Park 1960-1961. The special collections department at the University of Oregon holds all the Letters Kesey wrote to his best friend Ken Babbs during these drug experiments at the Stanford VA hospital Research program in Menlo park run by Dr. Leo Hollister. He tells Babbs detailed reports about his experiences on Drugs like AMT, LSD, Psilocybin, Mescaline, Ditran and some truly obscure molecules listed by three sets of different lettered Acronym’s. The EA-series, the JB-series & the IT-series drugs.
I looked into what these acronyms represented & found out EA is simply Experimental Agent (not Edgewood Arsenal) initially I couldn’t figure out the JB acronym so I asked Jay Stevens if he knew what JB represented. He thought it represented molecules that NASA was interested in looking at. I recently discovered what that JB acronym stands means. Its a Chemist, the initials of his first & last name--the JB series stands for Chemist ‘John Biel’ who Hollister knew very well. IT likely represents investigational Tryptamine, though I could be off on that.
Thought I would share this tid bit of info.