Shulgin’s Essential Amphetamines — Part 2
Biosynthesis and chemistry of the essential amphetamines...
In the last post I discussed Alexander Shulgin’s hypothesis that the molecular components of nutmeg essential oil — myristicin, safrole, and elemcin — might be transformed inside the liver to the psychedelic amphetamines, MMDA, MDA, and TMA. Although the evidence that this transformation occurs in humans, and thus that these amphetamines are responsible for nutmeg’s psychoactive effects, is rather weak, it leads to some rather interesting psychedelic drug chemistry, which is what I want to discuss in more depth in this post. In next weeks’ post, I’ll discuss the pharmacology of the psychedelic amphetamines, and how they compare to the classic psychedelics, in more detail.
After presenting his initial hypothesis, Shulgin soon noticed that, beyond the three components of nutmeg oil we already discussed, a range of similarly structured essential oil components (of various different plants) could potentially be converted to psychoactive amphetamines. This group of eleven amphetamines he referred to as his ‘essential amphetamines’ (i.e. amphetamines derived from essential oils), and each is given its own entry in PIHKAL (listed at the end of this post).
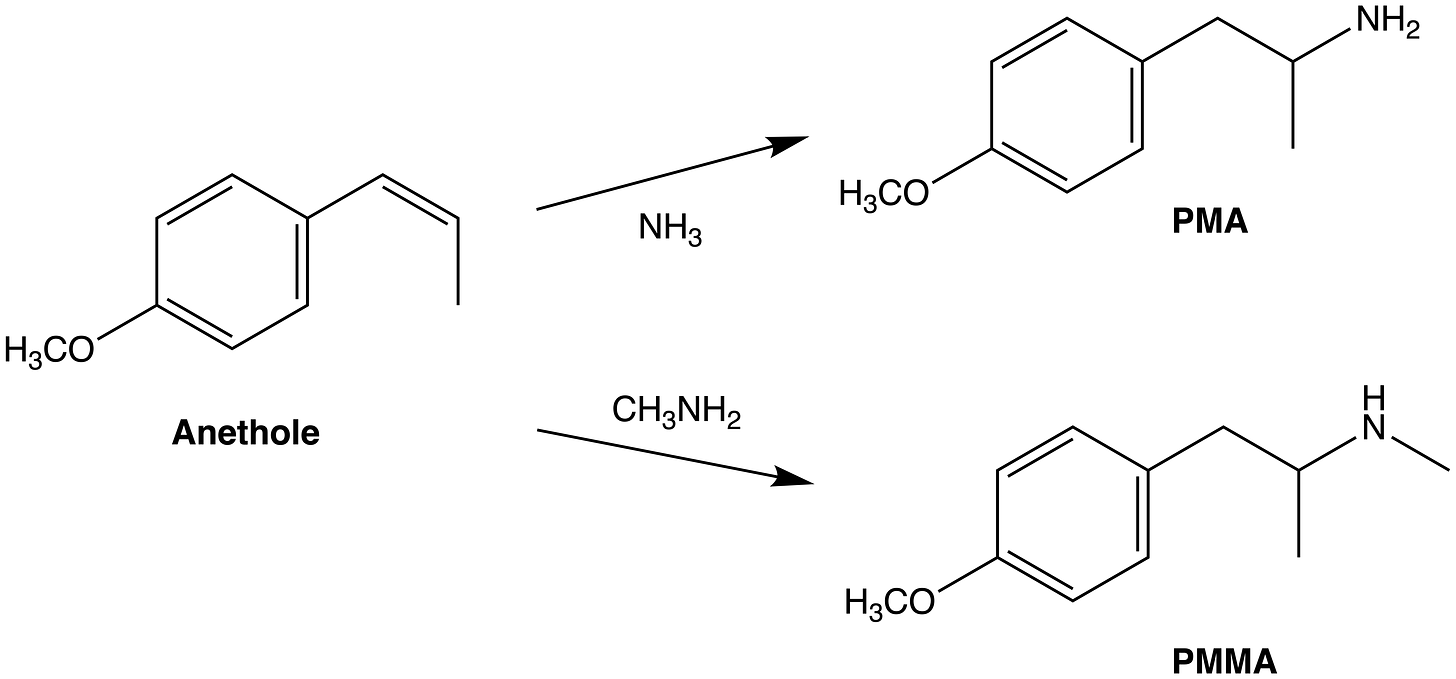
Each of the essential amphetamines can be formed in a single-step from its corresponding essential oil component (which I’ll refer to as the essential precursors) by the addition of an ammonia (NH3) molecule across the side chain double bond. Alternatively, addition of methylamine (CH3-NH2) instead of ammonia yields the methamphetamine derivative. Compare the syntheses of MDA and MDMA from safrole (the same basic reaction can be performed with any of the essential precursors):
The basic molecular skeleton of all ten essential precursors is derived from the amino acid phenylalanine, which is converted through several steps to the first of the essential precursors, anethole. This process involves removal of the amine (NH2) and carboxyl (COOH) groups from the side chain to yield a 3-carbon chain with a single double bond. The position of this double bond can vary, being either at the very end of the chain or shifted one position further towards the phenyl ring. However, this is of little significance when it comes to conversion to the amphetamine by addition of ammonia. Hydroxylation (addition of OH) and methylation (addition of a methyl group, CH3) of the phenyl ring at the 4-position yields anethole, which is found in anise and fennel oil and has a distinctly aniseed scent.
Addition of ammonia across anethole’s double bond yields 4-methoxy-amphetamine, which is better known as para-methoxy-amphetamine (PMA). As with safrole, addition of methylamine can also be performed to yield the methamphetamine derivative.
Note the numbering system of the phenyl ring carbons, starting from the carbon attached to the side chain (carbon 1) and moving sequentially around the ring (2 to 6). Carbon 4, which is directly opposite carbon 1, is often referred to as the ‘para’ position. This numbering system is important in describing and understanding the differences in the molecular structures.
From anethole, there remain four phenyl ring carbons to which oxygen-bearing groups can be attached. Basically, three things can happen at each carbon (at least in this context), depending on the expression of the relevant biosynthetic enzymes in the particular plant species:
Nothing. The carbon isn’t modified.
Hydroxylation and methylation to form a methoxy (OCH3) group.
Formation of a 5-membered methylenedioxy ring between two neighbouring carbons.
The methylenedioxy ring is particularly interesting, since it’s most recognisable as the ring structure in MDMA. It can be formed when the methoxy group methyl carbon is connected to the hydroxyl oxygen of a neighbouring carbon.
Whether or not this ring forms depends on whether or not a particular plant species expresses the relevant enzyme required for this chemical transformation. The same applies to the formation of the methoxy groups: the hydroxylase and methyl transferase enzymes that perform these transformations are selective and depend on plant species. Just because a phenyl ring carbon can be hydroxylated doesn’t mean it will be. This is why we see these different patterns of methoxy and methylenedioxy groups in different plant species.
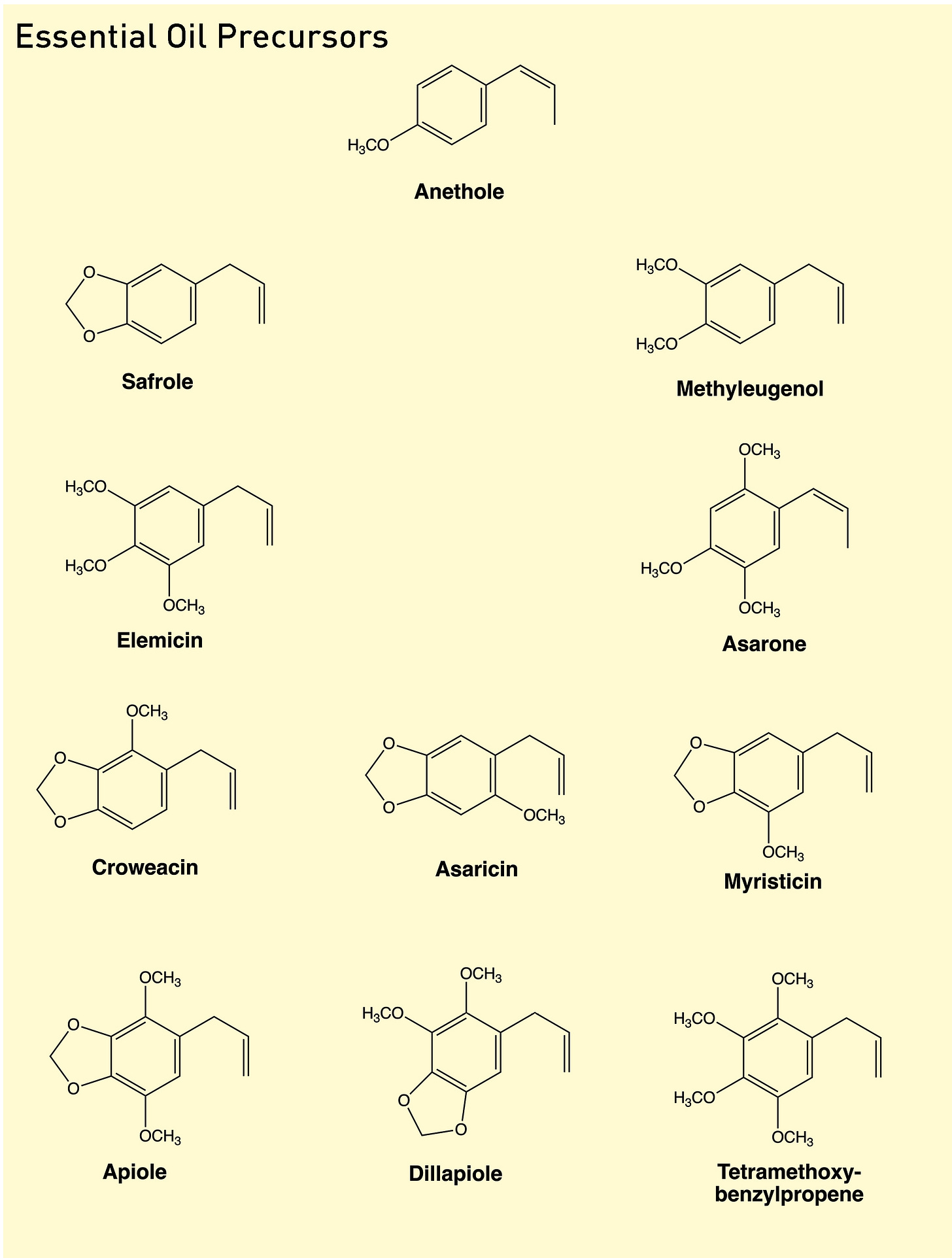
From anethole, there are a further ten ring patterns that Shulgin identified as essential oil components, giving us eleven essential precursors in total with the number of phenyl ring oxygens ranging from one to four:
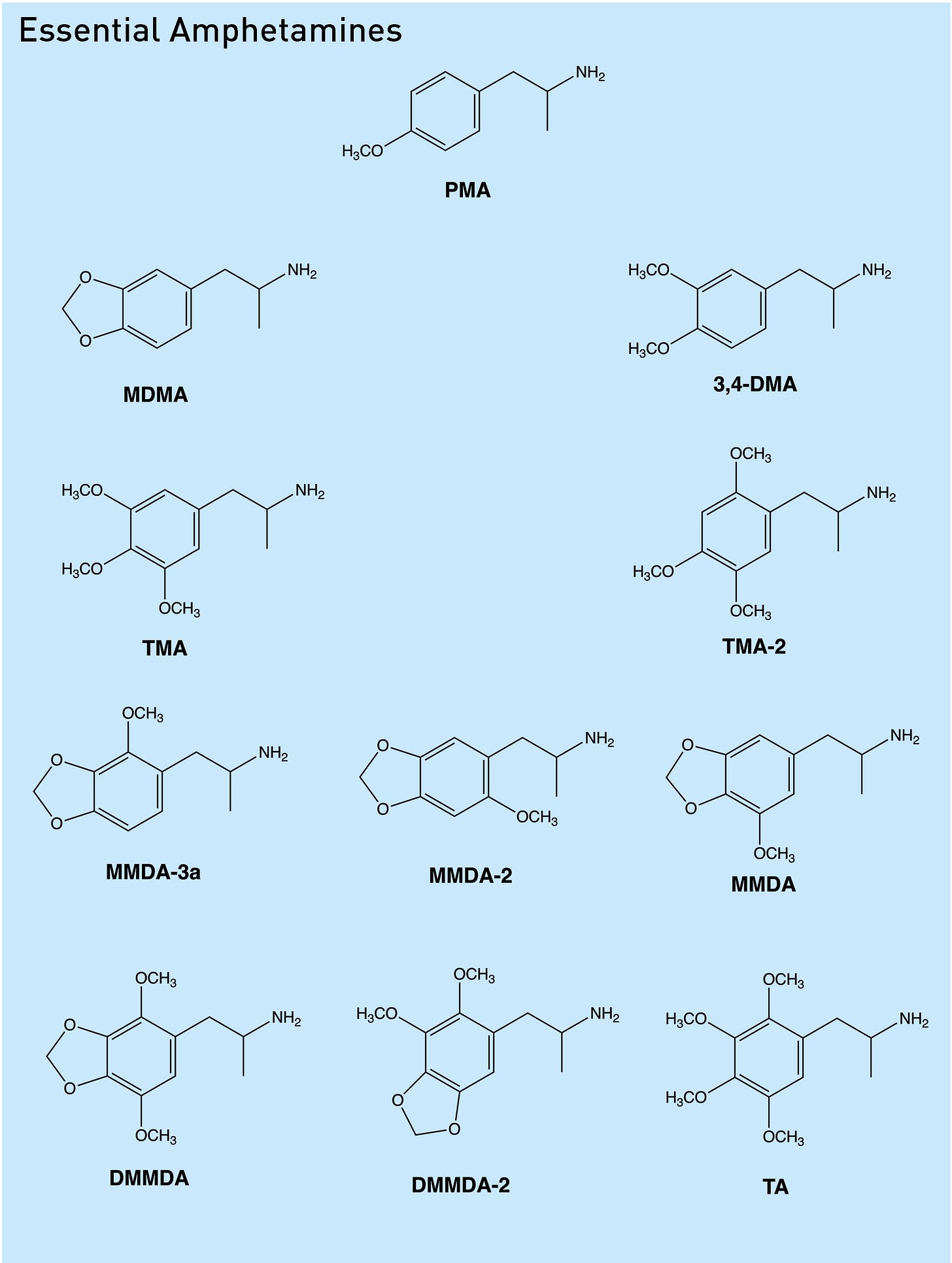
Here are the corresponding essential amphetamines:
Obviously, of the essential amphetamines, MDA and its methyl analogue, MDMA, are the most well-known. Since they are easily derived from the essential precursor safrole, sassafras oil (containing 70-80% safrole) has for decades now been an internationally controlled drug precursor with strict regulations on its purchase, import, and export. In 2008, the Cambodian government, with the help of the Australian Federal Police, made a show of burning 1278 barrels of the oil in their efforts to curb illicit MDMA manufacture. Whether or not this public display had any significant effect on global MDMA production, obtaining safrole for MDMA synthesis became prohibitively difficult for underground chemists, many of whom switched to the more readily available anethole, the precursor to PMA and PMMA — with tragic consequences.
PMA first appeared on the black market as a recreational drug in the early 1970s, reportedly sold under the moniker “chicken powder”. Its use dropped markedly until the 1990s, when it appeared again as drug manufacturers began making the switch from safrole to anethole, driven by drug precursor regulations. Unfortunately, this time, rather than being marketed as PMA/PMMA (which had a deservedly bad reputation following a number of well-reported deaths), it was often mis-sold as MDMA. This lead to a dramatic increase in deaths caused by the drug. In 2013, in the UK alone, PMA/PMMA caused just under 30 deaths. Fortunately, it has now largely disappeared from the market (unless you specifically seek it out).

PMA is toxic at much lower doses than MDMA, so users were often consuming what they thought was a standard dose of MDMA, but what was in actuality a toxic overdose of PMA. Compounding the problem, PMA’s effects take around two hours to manifest (at least twice as long as MDMA), so users were often doubling up when they experienced no effects after an hour or so. Prof. David Nutt explains the consequence of this:
"Their major problem is that they block the actions of the brain enzymes that offset the desired effects of serotonin and dopamine release that PMA/PMMA produce. This then massively accentuates their toxicity as the brain can’t compensate for the increase in serotonin so users can develop serotonin syndrome. This is a toxic reaction that elevates body temperature to a dangerous, and in some cases lethal, level.” (source)
The perennial advice applies: Test your drugs.
Shulgin’s speculations regarding myristicin and nutmeg have been extrapolated to other plant essential oils, with the claim that the essential precursor will be converted inside the body to the corresponding psychedelic amphetamine. However, again, there is very little evidence that this biotransformation occurs in humans. For example, extracts of Sweet Flag (Acorus calamus) have been marketed (and still are to some extent) as a “mild psychedelic”, often with the Shulgin-inspired claim that the asarone molecule converts to TMA-2 in the liver following ingestion. Marketing of calamus in this way seemed to reach a peak in the early 2000s, when large numbers of would-be-trippers found themselves in the ER with sustained (up to 15 hours) periods of severe vomiting after consuming the plant (link). This effect has nothing to do with TMA-2, but results from a powerful emetic effect of asarone itself. Best to avoid this one.
Most of the other essential amphetamines have at least some degree of psychoactivity, but some are more interesting than others. If you’re interested in exploring further, here is the complete set of essential precursors and their corresponding essential amphetamines, together with a link to the PIHKAL entry on the Isomer Design website.
1. The 4-methoxy precursor (anethole)
A distinctly aniseed scented molecule found in anise and fennel oil. Precursor to PMA
2. The 3,4-methoxy pattern (methyleugenol)
This clove-scented molecule is itself a mild local anaesthetic and converts to 3,4-dimethoxy-amphetamine (3,4-DMA) with addition of ammonia.
3. The 3,4-methylenedioxy precursor (safrole)
Found in nutmeg (as discussed in the last post) and sassafras oil. It’s safrole that’s responsible for the smell and taste of root beer. Precursor to MDA.
4. The 3-methoxy-4,5-methylenedioxy precursor (myristicin)
The highest concentration is found in nutmeg, but also present in black pepper, anise, and other plants. Precursor to MMDA.
5. The 2-methoxy-3,4-methylenedioxy precursor (croweacin)
Found in various Rue and Citrus species. Precursor to MMDA-3a.
6. The 2-methoxy-4,5-methylenedioxy precursor (asaricin)
An antifungal and insecticidal component of the oil of Ocotea opifera. Precursor to MMDA-2.
7. The 3,4,5-trimethoxy precursor (elemicin)
Component of the elemi tree (Canarium luzonicum), native to the Philippines, from which it gets its name. Precursor to TMA.
8. The 2,4,5-trimethoxy precursor (asarone)
Found in the Sweet Flag (Acorus calamus) and plants of the genus Asarum (from which it gets its name). Precursor to TMA-2.
9. The 2,5-dimethoxy-3,4-methylenedioxy precursor (apiole)
Found in parsley seed oil. This is the first precursor with four oxygen substituents around the phenyl ring. Precursor to DMMDA (dimethoxy-MDA) (PIHKAL 58)
10. The 2,3-dimethoxy-4,5-methylenedioxy precursor (dillapiole)
This precursor is a variation of the previous precursor, in which the methylenedioxy ring forms between two different neighbouring carbons, and is found in dill oil. Precursor DMMDA-2.
11. The 2,3,4,5-tetramethoxy precursor.
Finally, we have the precursor containing four oxygen substituents but with no methylenedioxy ring. A minor component of parsley oil. Precursor to tetramethoxy-amphetamine (TA)