Do Different Psilocybe Mushrooms Produce Different Effects? — Part 3
To the tryptamines and beyond...
Wrapping up this little trilogy of posts on a possible Psilocybe mushroom entourage effect, what has become clear to me, having spent quite a bit of time thinking about and discussing this topic over the last couple of weeks, is the strong conviction held by many very experienced users that different Psilocybe species reliably induce quite distinctive and characteristic effects. I’m certainly not one to dismiss the claims of those far more experienced with Psilocybe mushrooms than myself and my purpose in writing this short series is not to “debunk” such claims, but to consider what their chemical and pharmacological basis might be. Of course, the interaction between molecules and human neurophysiology is a richly complex topic and we shouldn’t be too disheartened when a satisfyingly compelling explanation fails to present itself.
See Part 2 here:
In the last post, I mentioned briefly that certain alkaloids, particularly baeocystin and norbaeocystin, might themselves be non-psychedelic (as strongly indicated by studies in animals) but, assuming they manage to cross the blood brain barrier, might modulate the effects of the known psychedelic components of the mushroom, psilocybin and psilocin. Whilst the primary locus of action of the classic psychedelics is the 5HT2A receptor, other serotonin receptors are involved in generating the psychedelic effect characteristic of a particular drug. The pattern of receptor stimulation by a molecule will determine the overall effect on the neuron and, consequently, on the behaviour of the networks they form. Around 16 serotonin receptor subtypes have now been identified, grouped into seven families:
The neurons within which these receptors are embedded are complex systems (in the technical sense): the neuron emerges from a multitude of interactions between a vast number of components. The receptors are the means by which a molecule external to the neuron is able to stimulate and perturb the complex intracellular network of signalling molecules that regulates the function and behaviour of the neuron.
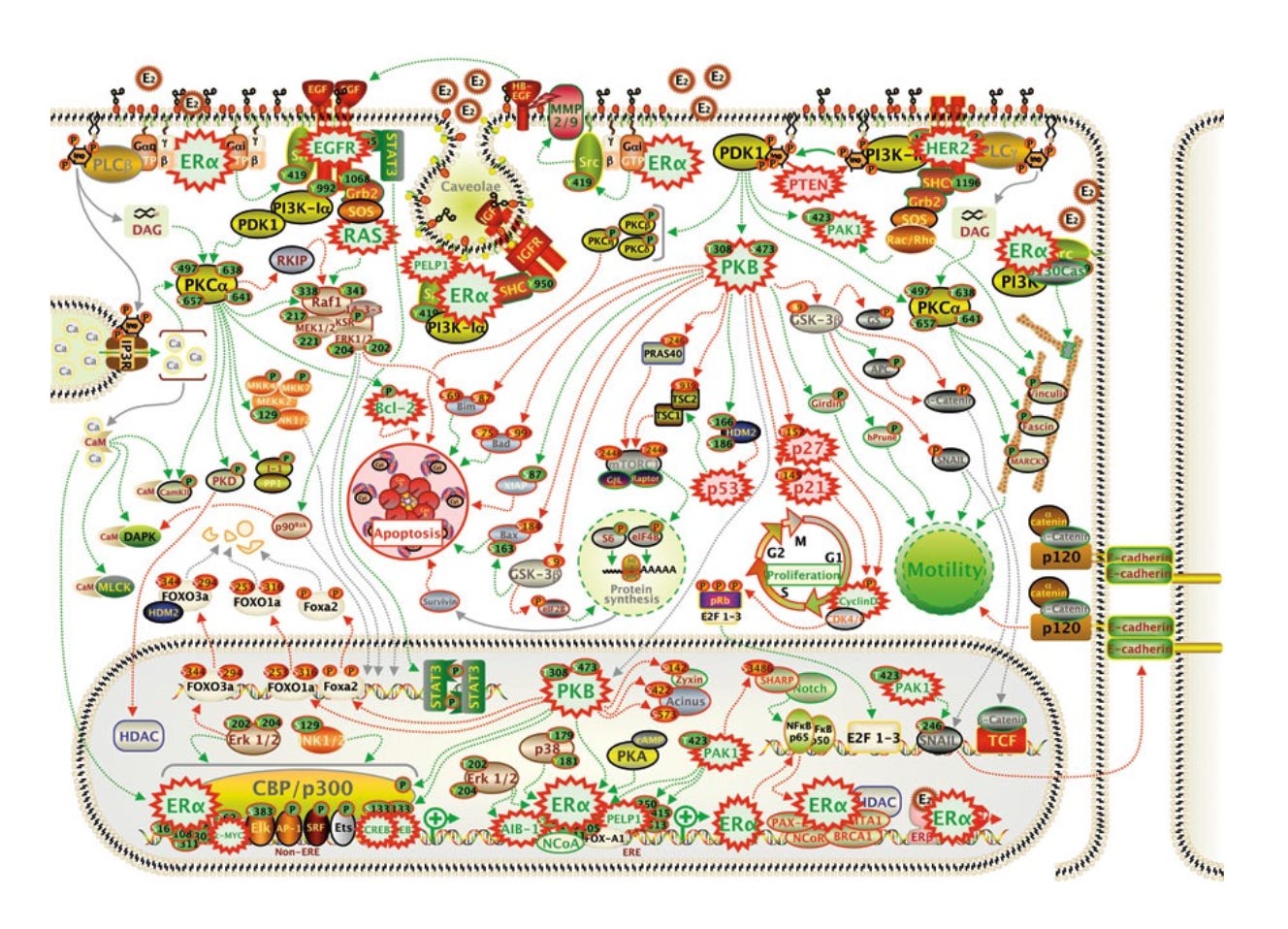
When multiple receptor (sub)types are stimulated by a single molecule, the overall effect on the neuron’s behaviour is extremely difficult to predict and, since the psychedelic effect is engendered by changes in the way millions of these neurons communicate and share information, so is the overall psychedelic effect. Using Sasha Shulgin’s musical analogy, a molecule doesn’t pluck a single string but, rather, plays a chord.
When multiple molecules interact with multiple receptor (sub)types, the picture becomes even more complicated: If a single molecule plays a chord, then multiple molecules are the sound of the orchestra. A molecule needn’t necessarily be psychedelic in its own right to contribute to the unique pattern of signalling elicited by a group of molecules and thus to modulate the subjective experience. We might call such non-psychedelic molecules psychomodulators (new term coined here!).
Beyond the beta-carbolines — including harmine and harmaline — that act as MAO inhibitors and allow DMT to be orally active, psychomodulators aren’t often discussed. Psychedelic drug combinations are obviously nothing new, but much less has been said about using non-psychedelic agents to modulate the experience elicited by psychedelic ones, aside from anxiolytics such as benzodiazepines to mitigate negative effects or antipsychotics as so-called trip killers. Having said that, I’m not as plugged into the underground drug forums as I used to be and I’d be interested to hear of any particularly interesting psychomodulators you might have come across in your clandestine explorations. I feel there is likely much low-hanging forbidden fruit here.
Speaking of beta-carbolines, a chemical analysis published in 2020 revealed their presence, somewhat surprisingly, in no less than five species of Psilocybe mushrooms (link). Beta-carbolines are most commonly associated with the ayahuasca vine, Banisteriopsis caapi, and the seeds of Syrian rue, Peganum harmala. Naturally, this attracted quite a lot of attention and, pertinently, prompted many to claim that perhaps the beta-carbolines are the secret to a Psilocybian entourage effect. Indeed, the authors of the study suggested:
“Psilocybe mushrooms produce an ayahuasca-like and potentially similarly synergistic set of metabolites that may impact upon onset and duration of their effects.”
This is quite a claim.
The detection of beta-carbolines in Psilocybe mushrooms is indeed surprising, but not entirely unexpected. Like the tryptamine alkaloid series more prominently expressed in magic mushrooms, the beta-carbolines (harmine and harmaline in particular) are derived from tryptophan, which is readily decarboxylated to tryptamine. As explained in detail in part 2, hydroxylation of the indole followed by double methylation of the amine nitrogen yields psilocin.
However, alternatively, the amine nitrogen of tryptamine can undergo a coupling reaction with the ubiquitous biochemical pyruvic acid (important in the energy-generating Krebs cycle). The product of this reaction then undergoes a highly favoured ring-closing reaction, followed by hydroxylation and methylation of the indole ring to generate harmaline. Loss of a hydrogen molecule (H2) then leads directly to harmine. Although this might appear somewhat complicated, the biochemical transformations are relatively trivial, so detecting these products in very low concentrations in mushrooms isn’t exactly miraculous.
As an interesting aside, the same ring-forming reaction is used by certain psychoactive cacti to convert phenethylamines, which include mescaline, into their double-ring isoquinoline analogues. Sasha Shulgin, most famous for PIHKAL (Phenethylamines I Have Known And Loved) and TIHKAL (Tryptamines I Have…), also threatened to pen a third installment, QIHKAL (Quinolines I Have…), covering analogues with the (iso)quinoline ring system. One of his final books, The Simple Plant Isoquinolines, is available (at a hefty price it seems), but doesn’t discuss their psychoactivity in the same manner as PIHKAL and TIHKAL — one for the serious chemists only I’d suggest.
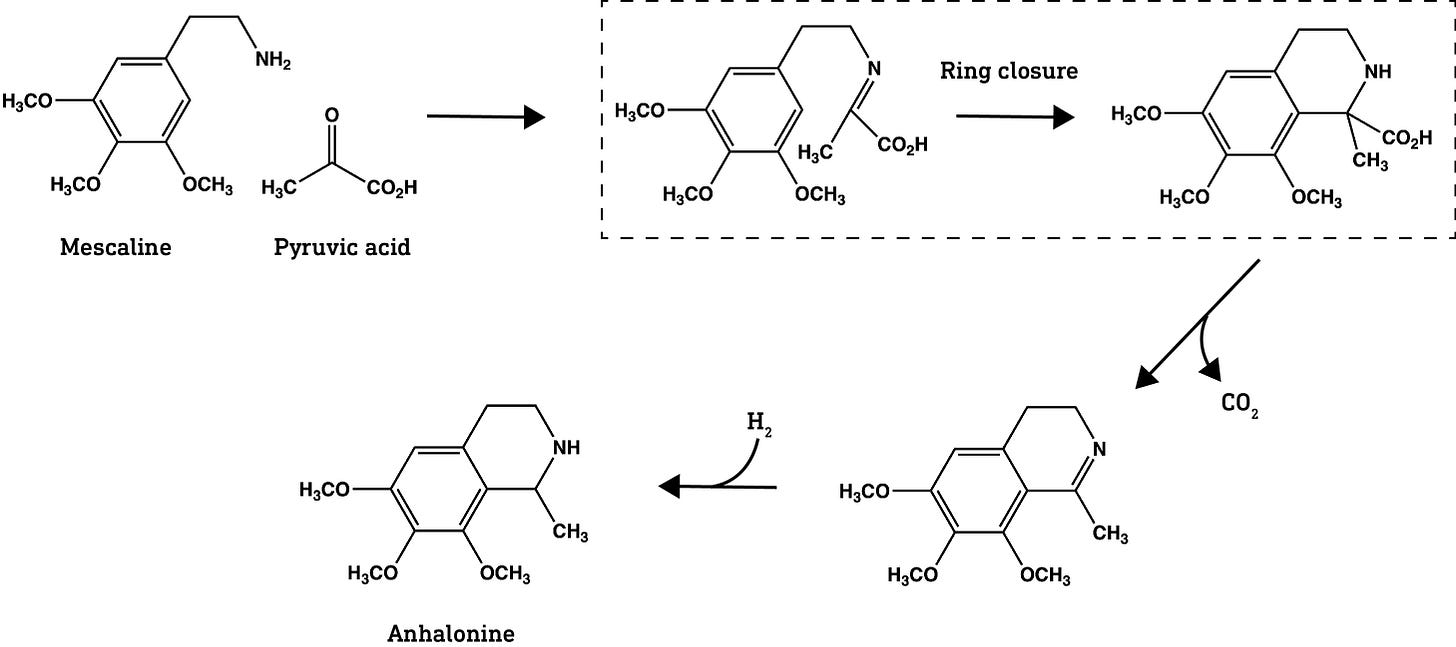
As a further aside, this ring-forming reaction is also used in the early steps of both morphine biosynthesis in opium poppies and in construction of the beautiful but deadly tubocurarine in the South American climbing vine Chondrodendron tomentosum (tubocurarine is the toxic component of the curare arrow poisons prepared using this plant). And I’m barely scratching the surface here — it’s really quite amazing what plants and fungi can build from a simple amino acid (one for a future post).
Anyway, back to the beta-carbolines. Without wanting to kill the buzz, almost all of the breathless commentary on this 2020 study failed to perform even a cursory quantitative analysis of the data reported in the paper. So let’s do that:
The concentration of beta-carbolines in the mycelia was measured at 21 ug/g, with concentrations in the mushrooms themselves being 100x lower. To put this into context, a 100ml dose of a typical ayahuasca decoction contains around 650mg of beta-carbolines. To achieve this dose using the analysed Psilocybe species would require consuming around 30kg of dried mycelia or 3 metric tons of the dried mushrooms.
A heroic dose if ever there was one.
So, whilst the discovery of beta-carbolines in Psilocybe mushrooms is certainly interesting from a biochemical perspective, the detected concentrations weren’t just low, they were analytical. There’s a saying in analytical chemistry that you can find any molecule you want in a sample if your equipment is sufficiently sensitive. Whilst this isn’t quite true, it’s certainly the case that, with modern instruments, it’s possible to detect vanishingly small quantities of metabolic side-products in biological extracts, many of which are likely to have little biochemical significance.
However, what this study does reveal is that there exists a complex chemical miscellany within Psilocybe mushrooms that we’ve only begun to explore. Whilst the psychoactive (and potentially psychomodulator) tryptamines are clearly the most prominent alkaloids of note, we can’t rule out the presence of other psychoactive molecules that we’ve yet to isolate and characterise. Plants and fungi are truly masterful chemists.
A recent chemical analysis using an advanced form of liquid chromatography coupled with mass spectrometry detected 1,465 unique molecular signatures in a sample of Psilocybe mushrooms, a chemical salmagundi from which only a handful could be assigned to known molecules (link). In other words, there’s an awful lot we don’t yet know about the chemistry of these magical little fungi and it’s certainly possible that their psychopharmacology might turn out to be just as complex.












So you seemed interested in knowing about other compounds that seem to modulate Psilocybin's action. I don't know if this qualifies but Piracetam is the best trip potentiator I've found, not only is the whole experience a lot more intense at the same dosage, it also seems to make my mind clearer, so the whole experience is more beneficial as I seem to get more depth of thought than with only the mushrooms which can make me quite fuzzy at times.
I'd tried mushrooms (and LSD) quite a few times in the past and didn't get any measurable effects until I tried them with Piracetam and only then did I understand what the psychedelic experience was. It just so happens that I've always had access to part of that experience/mindset without any substances since early childhood, so my first experiences were underwhelming because I was still close to my baseline. At any rate, I usually take 1-2g Piracetam with my mushrooms but I've heard that it also works with LSD, MDMA and probably most if not all tryptamines. I'd be very curious to read your explanation on this if you ever write about it as I don't properly understand how Piracetam works but it's one of my favourite compounds!
"A heroic dose if ever there was one." - rightfully deserving of its own paragraph.
The beta-carboline entourage effect argument seems a little bit like the Cannabis terpene entourage arguments. You'd need gargantuan amounts of cannabis to have enough terpenes to enact psychoactive effects. Maybe we'll see a follow up article in the Journal of Applied Homeopathy :P